If KPIs are not formally used, then asking the process owner how they know their process is effective is another good line of questioning for the internal audit checklist. So, if you have key performance indicators (KPIs), and these are maintained by your process owners, an assessment of process effectiveness can be included on your internal audit checklist by reviewing the KPIs and determining if the measures are showing that the process is meeting the expected outputs. This concept is for the process owner to have one or several main measures for their process that will let them know that the process is functioning as expected. Many companies will use the concept of key performance indicators for the processes when satisfying the ISO 13485:2016 requirements to evaluate performance. The second part of the ISO 13485:2016 internal audit requirements can be trickier to evaluate but, depending on the process, implementation can also be quite simple. How can you tell if the process is effective? Remember that the checklist is a tool for the auditor, and not something to give the auditee to fill out, so whatever format or questions and statements will be useful for the auditor to make sure that all important parts of the process are checked will work.
The checklist can include more than just questions it can also include statements from the procedures that the auditor wants to check. For the example above, the audit checklist could include questions on supplier evaluation, and a review of the supplier audit reports that have been collected, to see if they are done when determined by the QMS. The checklist is created by reviewing the ISO 13485:2016 standard and any documented procedures or undocumented processes for the activity to determine what should happen. An audit checklist is basically a set of questions that the auditor wants to ask, or activities that the auditor wants to witness, in order to verify the planned arrangements as above. The company might also specify that this is done using an audit of the customers every three years, which would be the company-defined criteria for the process.įrom this, we can start to create the audit checklist. As can be seen above, there are two sets of planned arrangements to check: those required by ISO 13485:2016, and those that the company has put in place for their process to function.įor example, if you are auditing a purchasing process against the ISO 13485:2016 standard (section 7.4.1), you will want to confirm that external providers are evaluated, selected, monitored, and reevaluated based on their ability to provide processes, or products and services, according to the requirements, and that their lack of commitment would affect their risk associated with the purchased product. So, when you are creating an audit checklist, you want to include the information needed to make sure that you successfully check these two outcomes of the process.Īn internal audit is there to witness the outcome of a process through a review of records or witnessing the actions of the employees, and then to compare this to the planned arrangements for the process to see if what is being done is what was planned. to make sure that the process is implemented and maintained effectively.
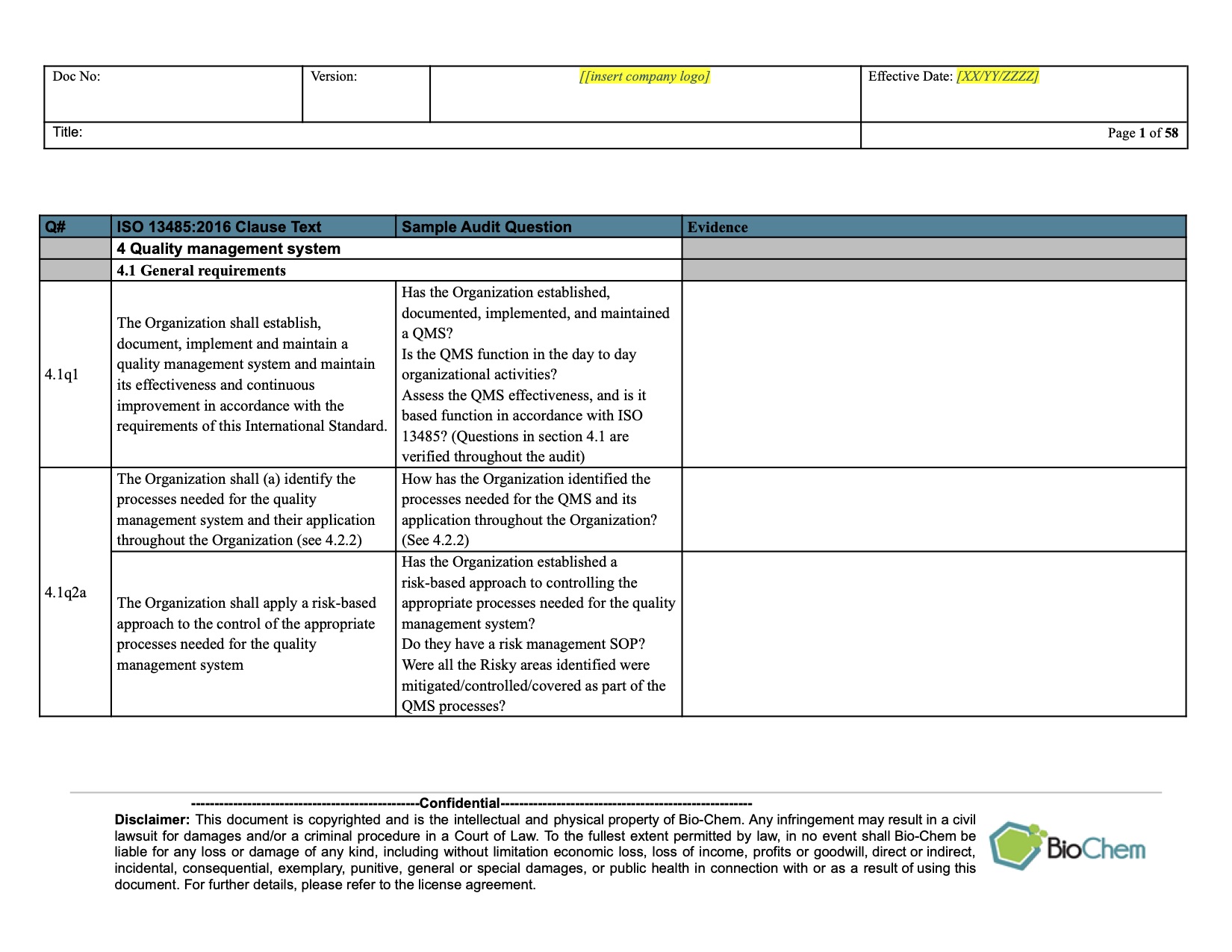

To better understand the why and how of internal audit checklists, it is helpful to understand what the ISO 13485:2016 requirements state about why we do internal audits. What does ISO 13485:2016 require the internal audit to do? Although audit checklists are not stated as a requirement in the ISO 13485:2016 standard, they are a widely used and important tool to make sure that when you perform an internal audit on a process, you do not miss any elements of that process. The ISO 13485:2016 requirements are very clear that this is a critical element of your QMS and, since you want to know how your processes are functioning, your internal audits become a key resource. One of the most important checking tools in a Quality Management System (QMS) for medical devices, or any management system, is the internal audit.
